Diabetes Technology Updates – Winter 2025
Nov 28, 2025
Diabetes technology is evolving at high speed.
Continuous Glucose Monitoring (CGM) and Automated Insulin Delivery (AID) are advancing in parallel, with new algorithms, new sensors, new hardware platforms, and new clinical indications appearing every few months. Keeping track requires systematic, continuous updates.
Below is a concise, structured overview of the most important developments across all major AID companies — including pivotal trials, real-world performance, hardware roadmaps, sensor compatibility changes, algorithm upgrades, and upcoming regulatory indications.
We cover:
- Medtronic Diabetes Care (MiniMed): MiniMed 780G
- Tandem Diabetes Care: Control-IQ
- Insulet: Omnipod 5
- mylife Diabetes Care: mylife Loop
- Diabeloop: DBLG1
- Beta Bionics: iLet Bionic Pancreas
- Medtrum: TouchCare Nano System
- Sequel Med Tech: twiist AID system
- Open-source AID systems (AndroidAPS, DIY Loop, iAPS, Trio)
Get Access To Updated Diabetes Technology Courses
If you'd like deeper training on these systems, all corresponding courses are available at diabetotech.com.
1. Medtronic Diabetes Care (MiniMed): MiniMed 780G

MiniMed 780G: Userbase and Performance
- Estimated Userbase: Estimated at >700,000 individuals although Medtronic has not shared its userbase since 2020.
- Pivotal Trial (n=157): TIR 75%, TBR 2.3%
- Real-World Data presented @ADA2025 (n=374 548): TIR 72%, TBR 1.4%
- Link to latest quarterly update
Sensor Updates

- The Instinct sensor has launched in the US for individuals with type 1 diabetes 7 years and older.
- No CE-mark yet; no FDA-approval for type 2 diabetes yet
- No news on extended wear or other expected Simplera updates
Hardware Updates
Upcoming Insulin Pumps:
- 800-Series Pump = MiniMed Flex:
- Smaller, tubed pump controllable via new Android and iOS apps.
- Compatible with existing Extended reservoirs and infusion sets.
- Expected Availability: 2026.
- Modular Patch Pump = MiniMed Fit:
- Slightly larger than Omnipod pods.
- Insulin Capacity: 300 units.
- Wear Duration: 7 days with Extended wear infusion set.
- Expected Availability: 2027.
No updates on
- MiniMed combined patch pump + sensor (MiniMed Duo technology)
- Implanted pump (MIIPS) - Phoenix study is ongoing: first implants in the Netherlands & France
Algorithm Updates
- SmartGuard™ Enhancements:
- Recent trials with Simplera sensor show TIR up to 80% in adults.
- Algorithm updates will be available via future software updates.
- Vivera = Novel Medtronic Experimental Automated Insulin Delivery (NMX-AID) = 3rd generation full closed-loop algorithm:
- Optional meal bolusing allows the system to bolus each meal automatically or users may choose to bolus using a simple meal announcement or a traditional carb announcement.
- Revolutionized hourly adaptation and personalization of insulin deliveries.
- Pump Start Feature: Bypasses 48-hour warm-up for SmartGuard activation.
- Pivotal Study: GATEWAY recruiting in US with MiniMed Flex, upcoming NEXUS study for outside US with MiniMed Flex.
Expanding Indications
- Preschoolers (2–6 years):
- CE-approved, FDA-submitted
- LENNY trial positive results published; SUCCEED2 trial results expected @ATTD2026
- Pregnancy:
- CE-approved, FDA-submitted
- CRISTAL trial positive results published
- Type 2 Diabetes:
- CE & FDA-approved for adults 18+ with insulin-requiring type 2 diabetes
- IMPACT2D trial positive results published
Newer Insulins
- Current Approval: NovoRapid®, NovoLog®, or Humalog® 100 U/mL.
- CE Approval: Fiasp® and Lyumjev®.
Want to learn more? Check out our updated course on the MiniMed 780G pump and AID system, Guardian 4, and Simplera CGM - Course on the Instinct CGM is coming soon!
#2 Tandem Diabetes Care: Control-IQ

Control-IQ™: Userbase and Performance
- Estimated Userbase: Approximately 320,000 users.
- Pivotal Trial (n=168): TIR 67%, TBR 2.1%
- Real-World Data (n= 20 000): TIR 71%, TBR 1.5%
- Link to latest quarterly update
Sensor Updates
- t:slim X2 Pump Compatibility:
- Dexcom G6 and G7
- FreeStyle Libre 2 Plus & 3 Plus: only available in the US
- Mobi Pump Compatibility:
- Dexcom G6 and G7
- FreeStyle Libre 3 Plus: Expected in 2026 in the US.
Hardware and App Updates
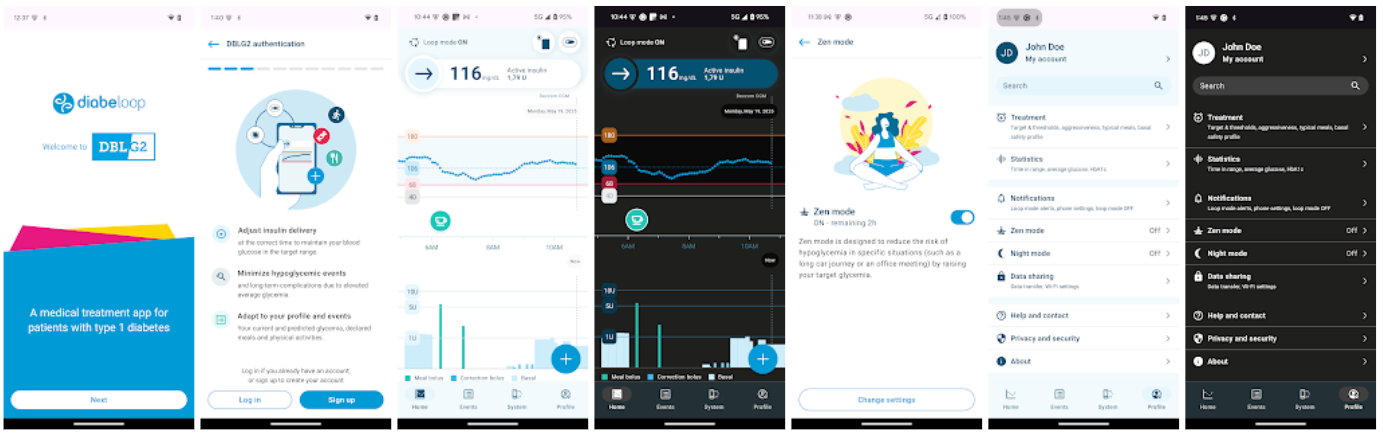
- t:slim X2 Pump:
- Tandem t:slim Mobile App (Android & iOS): Includes phone bolus feature (only in US, New Zealand & Australia), coming to Europe “soon” together with the dedicated Tandem Source readout platform.
- Mobi Pump:
- Tandem Mobi Mobile App: available for iOS users in the US, recent FDA-approval for Android version, rollout in 2026 in the US
- European Launch: Unclear.
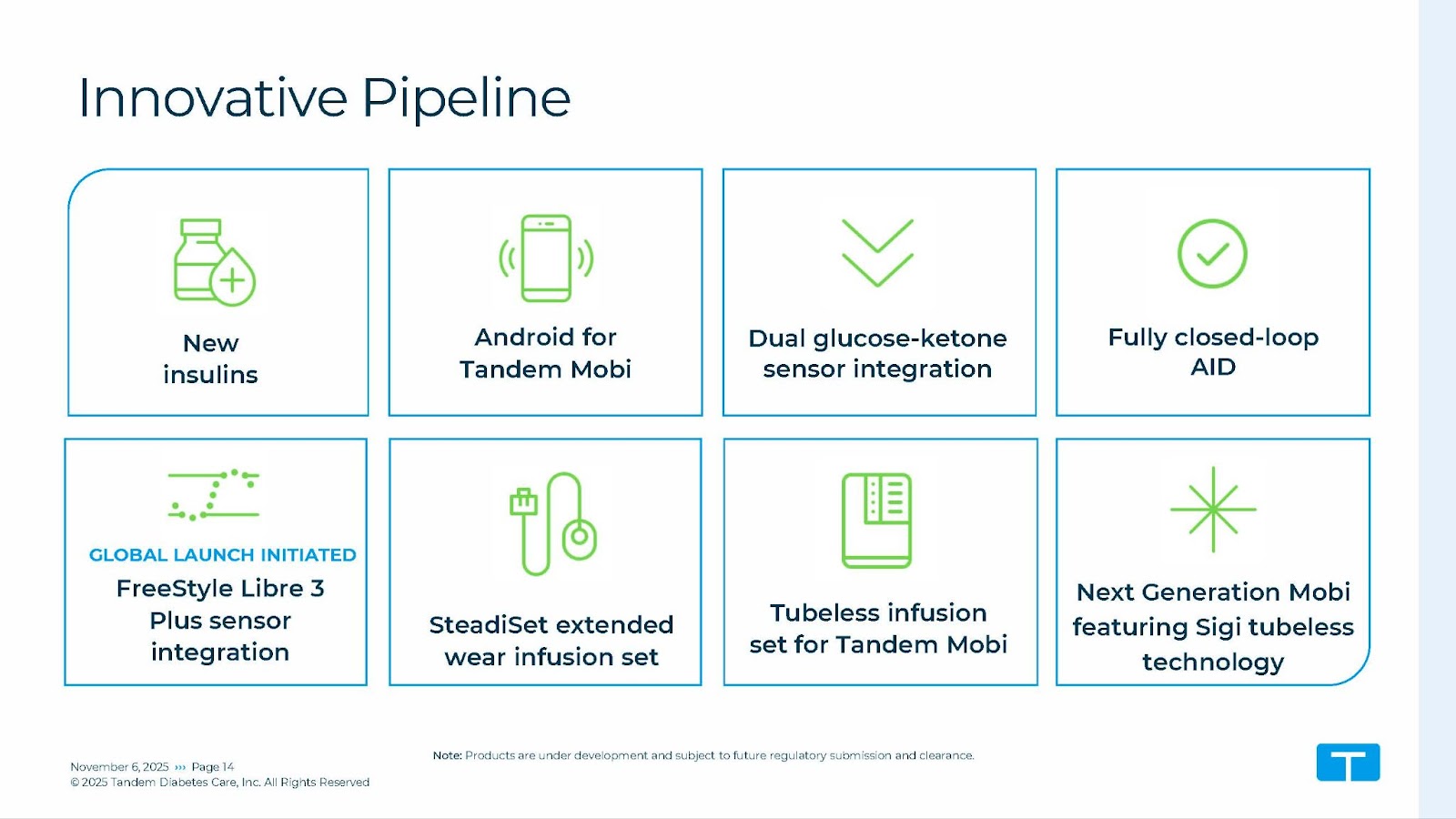
- Future Developments:
- 7-day wear SteadiSet Infusion Set: US-launch is expected in 2026
- Mobi:tubeless: expected launch 2026 in US
- Sigi Pump: Under development.
- No updates on the t:slim X3 pump.
Algorithm Updates
- Control-IQ+ Upgrade
- Expanded Insulin Dose Range (10-100 >> 5–200 U/day) & Weight Range (25-140 >>> 9-200 kg/day).
- Includes new onboarding wizard: automatic system settings after entering weight and total insulin dose in the wizard
- Launched in US, European launch unclear
- Freedom fully closed-loop Algorithm
- AIDANET fully closed-loop Algorithm
- Feasibility trial (n=6) positive results published
- Feasibility trial (n=34) positive results published
- Randomised trial ongoing in adults and pediatrics
Expanding Indications
- Preschoolers (2-6 years):
- FDA-approved, CE-submitted
- PEDAP trial positive results published
- Pregnancy:
- No FDA or CE-approval yet
- CIRCUIT trial positive results published
- Type 2 Diabetes:
- FDA-approved, CE-submitted
- 2IQP trial positive results published
Newer Insulins
- Current Approval: NovoRapid®, NovoLog®, or Humalog® 100 U/mL.
- European Approval: Lyumjev®, based on positive trial results.
Interested in updates? Explore our courses on the Tandem t:slim X2 pump & AID system, and the Mobi pump & AID system.
#3 Insulet: Omnipod 5
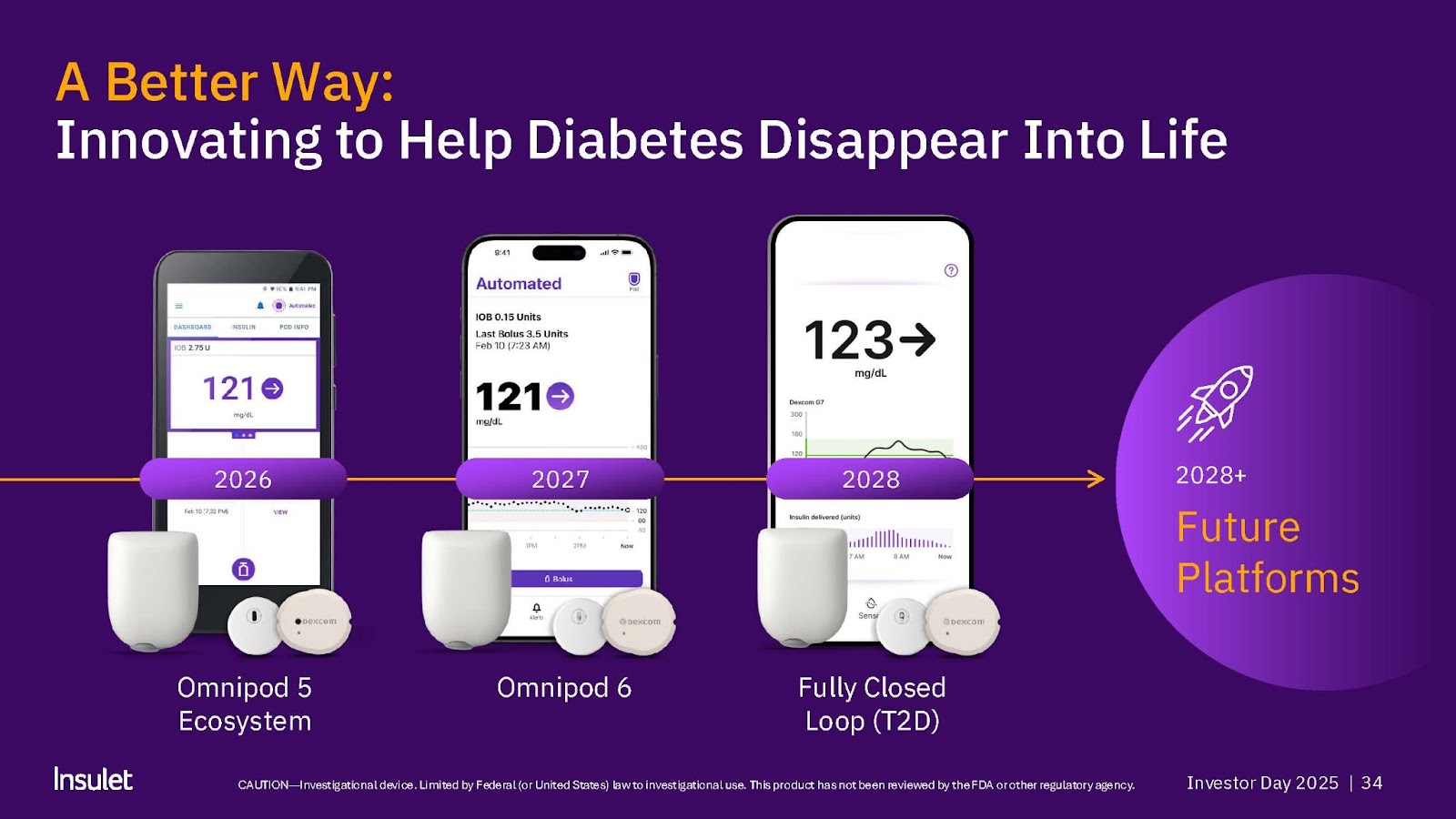
Omnipod 5: Userbase and Performance
- Estimated Userbase: Approximately 365,000 users.
- Pivotal Trial (n=235): TIR 74%, TBR 1.3% in adults
- Real-World Data (n=69 902): TIR 69%, TBR 1.1% with glucose target 110 mg/dl
- Link to latest quarterly update
Sensor Updates
- Compatibility:
- Dexcom G6 & G7
- FreeStyle Libre 2 Plus in select European countries
- FreeStyle Libre 3 Plus expected launch in 2026
Product Updates
- Geographical Expansion
- Current Markets: U.S., Germany, UK, Netherlands, France, Australia, Belgium, Canada, Israel, Italy, Switzerland, Nordics.
- Upcoming launches: Kuwait, Saudi Arabia, UAE, Qatar, Spain.
- Omnipod 5 app
- Omnipod Discover
- Insulet’s dedicated readout platform with weekly insights to identify patterns
- Recently launched in US
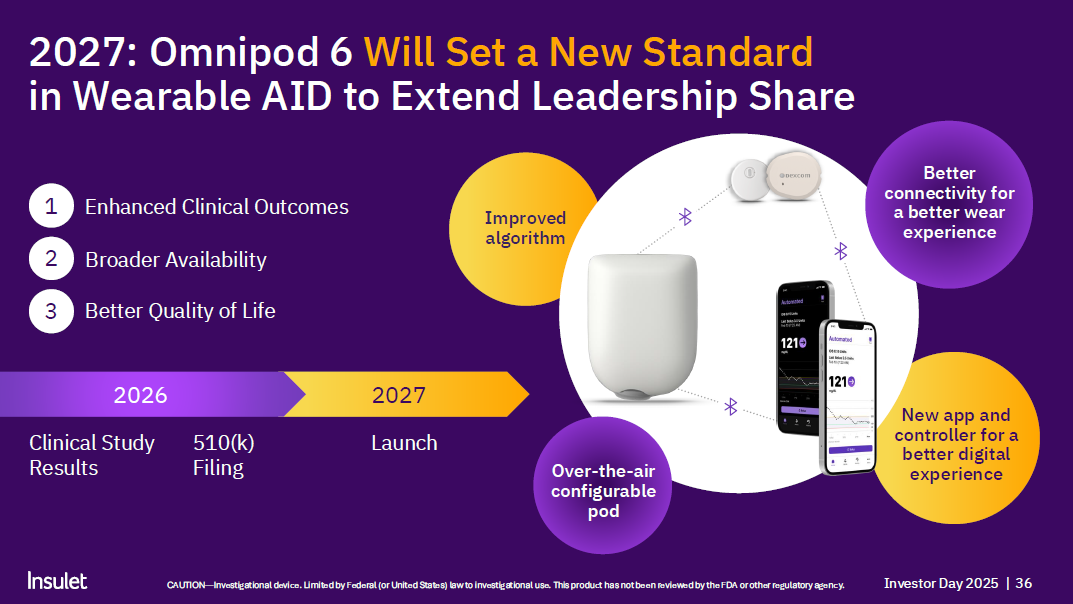
- Omnipod 6
- Improved algorithm
- Better connectivity for a better wear experience
- New app and controller for a better digital experience
- Over-the air configurable pod
- Expected launch 2027
- Fully closed-loop AID for people with type 2 diabetes
- Ready out-of-the-box: designed for self-start
- Designed for ease for HCPs to prescribe with no settings to start
- Expected launch 2028
- Expected updates after 2028
- Fully closed-loop AID for people with type 1 diabetes
- Higher insulin capacity
- AID for people with type 2 diabetes on basal-only insulin
- Longer wear time
- Next-gen pod hardware platform
No updates on Omnipod Go (FDA-approved basal-only insulin pod for people with type 2 diabetes).
Algorithm Updates
- SmartAdjust 2.0:
- Enhancements: 100 mg/dl (5.6 mmol/l) target glucose, more time in auto mode and improved algorithm responsiveness.
- Expected launch 2026
- Feasibility trial (n=75) ongoing in New Zealand, STRIVE trial (n=132) ongoing in US, results will be presented @ADA2026
- EVOLUTION (fully closed-loop) Algorithm:
- EVOLUTION trial positive results published
- EVOLUTION2 trial is ongoing, results expected @ATTD2026
- SIMPLEUSE trial is studying the optimal settings for the full closed-loop system in people with type 2 diabetes
- EVOLUTION3 feasibility trial will start end of 2025
- Pivotal full-closed loop trial will start in 2026
Expanding Indications
- Preschoolers (2-6 years):
- FDA- & CE-approval
- Pivotal trial positive results published
- Pregnancy:
- Off-label use - no official approval planned, no trials running in pregnancy
- Type 2 Diabetes:
- FDA-approved, CE-submission expected in 2026
- SECURE-T2D positive results published
Newer Insulins
- Approved Insulins: NovoRapid®, NovoLog®, Trurapi®, Kirsty®, Humalog®, Admelog® 100 U/mL.
- Off-Label Use: Reports of Humalog® 200 U/mL usage; lacks supporting data and constitutes off-label use.
Want to dive deeper? Check our updated course on the Omnipod 5 pump and AID system.
#4 mylife Diabetes Care: mylife Loop
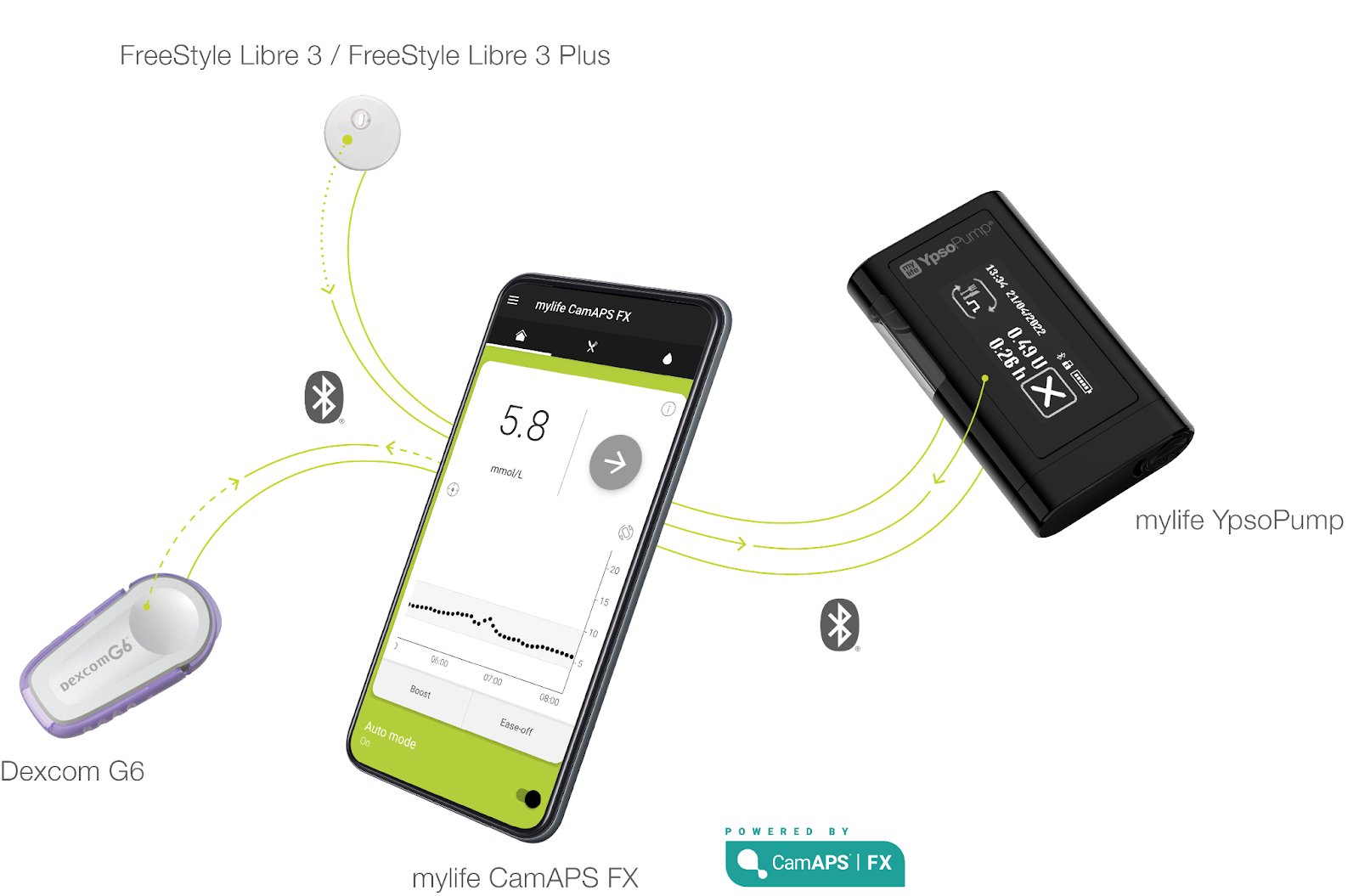
Userbase and Performance
- Estimated Userbase: Approximately 75,000.
- Pivotal Trial (n=86): TIR 64%, TBR 2.6%
- Real-World Data (n=1805): TIR 73%, TBR 2.3%
- Private company, no investor updates available
Sensor Updates
- Compatibility:
- Dexcom G6
- FreeStyle Libre 3 & 3 Plus
- Future: Dexcom G7 - no timeline available
Product Updates
- Geographic availability
- Available in most European countries, UK, Canada, New Zealand & Australia
- US: the mylife YpsoPump is submitted to the FDA, the CamAPS FX algorithm is FDA-approved
- mylife CamAPS FX app
- Available for Android & iOS
- Companion feature recently also launched for iOS devices
- mylife YpsoPump reservoir
- Because of a structural shortage of NovoRapid PumpCarts, mylife Diabetes Care now promotes their refillable reservoir more prominently. Their reservoir can be refilled once within 7 days after the initial filling process.
- mylife Cloud
- Dedicated readout platform is in development (current integration with Glooko) - no timeline available
- Patch pump in development - no timeline available
Algorithm Updates
- CamAPS HX (full closed-loop) algorithm:
- CE-label for people with type 2 diabetes
- Available on iOS and Android, but not commercially available
- CLEAR trial positive results published in n=24 adolescents and n=26 adults with type 1 diabetes

Expanding Indications
- Preschoolers (2-6 years)
- CE & FDA-approved
- KidsAP02 trial positive results published
- Pregnancy
- CE & FDA-approved
- AiDAPT trial positive results published
- Type 2 Diabetes:
- CamAPS HX is CE-approved for people with type 2 diabetes
- Small trial (n=26) with CamAPS HX positive results published
- COYOTE trial with CamAPS HX ongoing, results expected in 2027.
Newer Insulins
- Approved Insulins: All U100 rapid-acting and ultra-rapid-acting insulins.
- Recent Studies: Lyumjev improves postprandial glucose excursions in case of a missed insulin bolus
Learn more: Access our courses on the mylife YpsoPump and mylife Loop system.
#5 Diabeloop: DBLG1

Userbase and Performance
- Estimated Userbase:
- Diabeloop-Insight: 10,000 (2022) will be phased out as Roche no longer supports the Accu-Check Insight pump
- Diabeloop-Kaleido: 4000 users in the Netherlands, Germany and France (source: DBLdata)
- Diabeloop-Dana: 80+ users, recently launched in October 2025
- Pivotal Trial (n=63): TIR 69%, TBR 2.0%
- Real-World Data (n=3706): TIR 72%, TBR 0.9%
- Private company, no investor updates available
Sensor Updates
- Compatibility:
- Dexcom G6
- Future: Dexcom G7 - integration in final phase
Product Updates
- Availability:
- Diabeloop-Kaleido: Netherlands, Germany, France.
- Diabeloop-Dana: Germany, extending to new geographies when G7 is integrated
- Future: possible combination with Terumo’s MEDISAFE WITH insulin pump.
- DBLG2 app
- Algorithm on Android app with improved user interface and nightmode
- iOS version to be released as soon as CE mark is granted, with Dexcom G7
- Recent limited release with Kaleido in Netherlands, launch planned in Netherlands & Germany in 2026. Launch with Dana-i pump early 2026.
- Kaleido 2 insulin pump
- Smaller version of the Kaleido pump with enhanced internal technology
- Expected launch 2026
Algorithm Updates
- Full closed-loop algorithm
- Timeline unclear
Expanding Indications
- Children:
- DBL4T trial in children 12-17 years finished, results not available
- Pregnancy:
- Off-label use - no official approval planned, no trials running in pregnancy
- Type 2 Diabetes:
Newer Insulins
- Approved Insulins: NovoRapid®, Humalog® 100 U/mL.
- Virtual Simulator Study: Safe use with Lyumjev® and Fiasp®; Lyumjev showed better TIR.
Interested in Diabeloop? Explore our courses on the Accu-Chek Insight pump, Kaleido pump, Diabeloop-Insight and Diabeloop-Kaleido system. Module on Dana-i pump coming soon.
#6 Beta Bionics: iLet Bionic Pancreas

Userbase and Performance
- Estimated Userbase: Approximately 24 000 users.
- Pivotal Trial (n=219): TIR 65%, TBR 1.8%
- Real-World Studies (n=16 394): HbA1c: 8.9% > 7.3%. TIR/TBR: Data not specified.
- Link to latest quarterly update
Sensor Updates
- Compatibility:
- Dexcom G6 and G7
- FreeStyle Libre 3 Plus
Product Updates
- Availability: U.S. only.
- Mobile Apps:
- iLet Mobile App: View-only for users (Android and iOS).
- Bionic Circle App: Dedicated follow app for Android.
- Mint Modular Patch Pump
- 200U insulin reservoir, 3-day wear time, steel needle (manual insertion)
- No need to connect to your phone during patch changes, no need to recharge
- Expected launch 2027
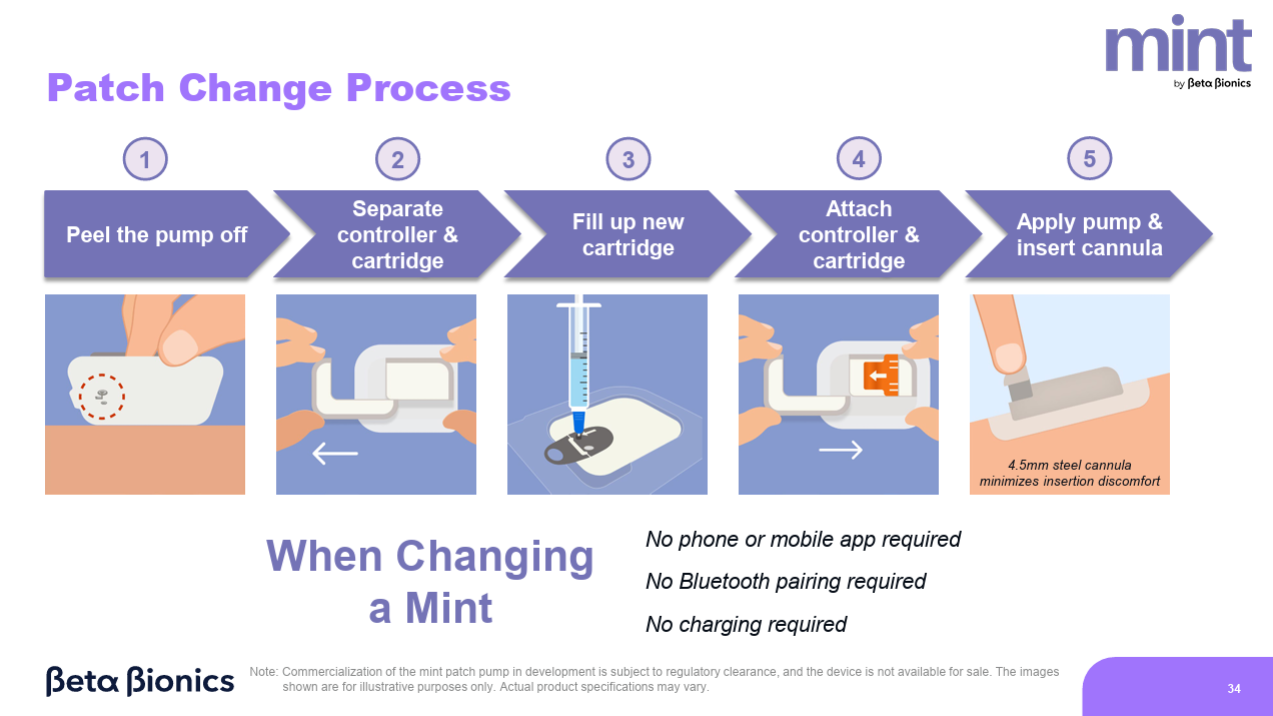
- Bihormonal Bionic Pancreas:
- Design: Uses prefilled glucagon cartridges and separate infusion set.
- Exclusive partnership for glucagon with Xeris.
- Feasibility trial: expected start end of 2025.
Algorithm Updates
- Insulin-Only Algorithm: No updates currently planned.
- Focus: Advancing bihormonal system.
Expanding Indications
- Preschoolers (2-6 years)
- Off-label use - no official approval planned, no trials running in preschoolers
- Pregnancy
- Off-label use - no official approval planned, no trials running in pregnancy
- Type 2 Diabetes
- Off-label use - 25% of new pump starts have type 2 diabetes
- Preliminary, unpublished studies show potential benefits.
Newer Insulins
- Approved Insulins: U100 NovoRapid®, Humalog®, Fiasp® PumpCart.
Want more information? Check out our course on the iLet Device and System.
#7 Medtrum: TouchCare Nano System

Userbase and Performance
- Estimated Userbase: Approximately 4000 users.
- No randomised trials available
- Real-world data:
- Small study (n=20) in Argentia positive results published
- Small study (n=10) in Italy positive results published
- Ongoing Study: Before-and-after study in France with 160 participants.
- Private company, no investor updates available
Sensor Updates
- Compatibility:
- TouchCare Nano CGM.
- Future: Medtrum S10 CGM (all-in-one device)
Product Updates
- Availability: most European countries + UK
- Pump Options:
- 200 U Patch Pump
- 300 U Patch Pump
- Future: 400 U Patch Pump - timeline unclear
Algorithm Updates
- APGO Algorithm: No available results or updates.
Expanding Indications
- Data Unavailable: For preschoolers, pregnancy, or type 2 diabetes.
Newer Insulins
- Approved Insulins: U100 NovoRapid®, Humalog®, Fiasp®, Lyumjev®.
Want more information? Check out our course on the TouchCare Nano Pump and TouchCare Nano System.
#8 Sequel Med Tech: twiist AID System

Userbase and Performance
- Estimated Userbase: Approximately 2000 users.
- Pivotal Trial Tidepool Loop (n=558): TIR 73%, TBR 2.8%
- Real-world data (n=19+35) presented @ADA2026: TIR 70-80%, TBR 2.9-3.0%
- Private company, no investor updates available
Sensor Updates
- Compatibility:
- FreeStyle Libre 3 Plus
- Future: Eversense 365
Product Updates
- Availability: US only.
- Twiist AID app: iOS only + twiist insiight follow-app for iOS
- Future Pump Options:
- More infusion set choice
- Dedicated on-body patch
- Availability for Android
Algorithm Updates
- Tidepool Loop 1 = current algorithm
- Tidepool Loop 2:
- Tidepool is working to get FDA-approval for an updated Loop algorithm
- Sequel Med Tech is open to integrate other algorithms if FDA-approved
Expanding Indications
- Data Unavailable: For preschoolers, pregnancy, or type 2 diabetes.
Newer Insulins
- Approved Insulins: U100 NovoLog®, Humalog®
- Co-development agreement with Arecor (ultrafast insulin 500 U/ml)
Want more information? Check out our course on the twiist Pump and twiist AID System.
#9 Open-Source AID Systems: AndroidAPS, DIY Loop, iAPS, Trio
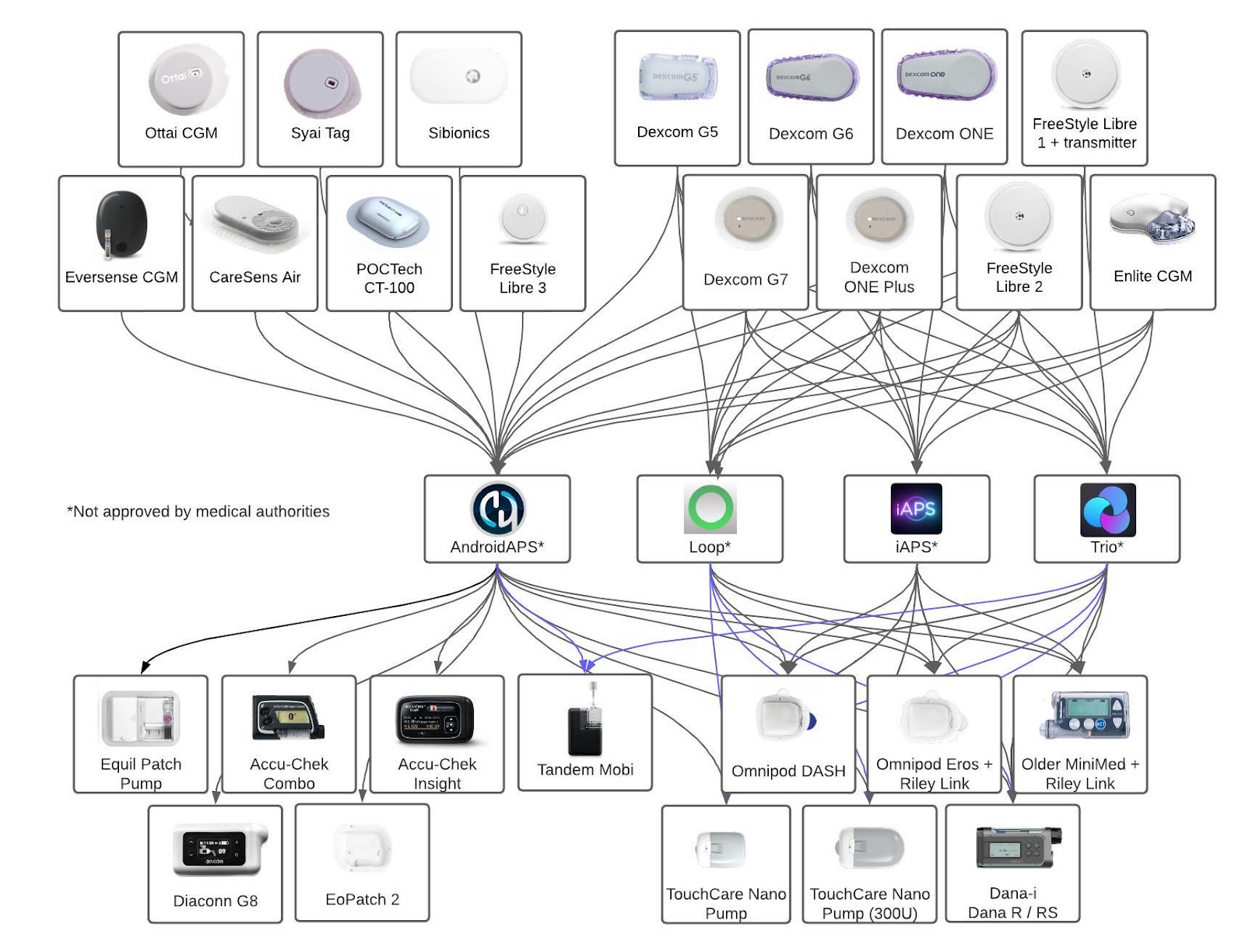
Userbase and Performance
- Estimated Userbase: Approximately 25,000 users.
- Key Studies:
- AndroidAPS RCT (n=44): TIR 71%, TBR 1.6% (New Zealand).
- AndroidAPS Real-World (n=142): TIR 79%, TBR 4% (Chine)
- DIY Loop Real-World (n=558): TIR 73%, TBR 2.9% (U.S.).
- Supported Open-Source AID (n=248): TIR 80%, TBR 2.5% (Canada).
- Ongoing Study: 500 participants in China.
Sensor and Pump Updates
- AndroidAPS v3.3.2.1: Released August 2025 - Tandem Mobi support “should be available” by end of 2025
- DIY Loop v3.8.2: Released October 2025 with Dana pump support - TouchCare Nano Pumps support is available in the developer branch
- iAPS v8.03.3: Released November 2025 with Dana and TouchCare Nano support; concerns over safety due to frequent updates.
- Trio v0.6.0: Released October 2025 with Dana support - TouchCare Nano Pumps support is available in the developer branch. Coming soon: support for Tandem Mobi & Eversense.
Algorithm Updates
- Full Closed-Loop: AndroidAPS supports unannounced meals.
- Pancres4All Trial: Demonstrated feasibility of AndroidAPS in full closed-loop mode over 3 days in 16 participants with type 1 diabetes.
- CLOSE IT Trial: 12-week use of Lotus in 37 participants with type 1 diabetes showed that full closed-loop mode was non-inferior to hybrid closed-loop mode (presented @EASD2025)
Results CLOSE IT trial presented by prof Neale Cohen @EASD2025
Expanding Indications
- Preschoolers: Usage reported; data lacking.
- Pregnancy: Limited literature but in use.
- Type 2 Diabetes: Reports show mean TIR of 93% in small group.
Newer Insulins
- Common Choices: Fiasp®, Lyumjev®.
- Insulin Mixing: Some users practice; limited data available.
Want more information? Check out our course on AndroidAPS, DIY Loop and iAPS. Module on Trio coming soon.
Conclusion
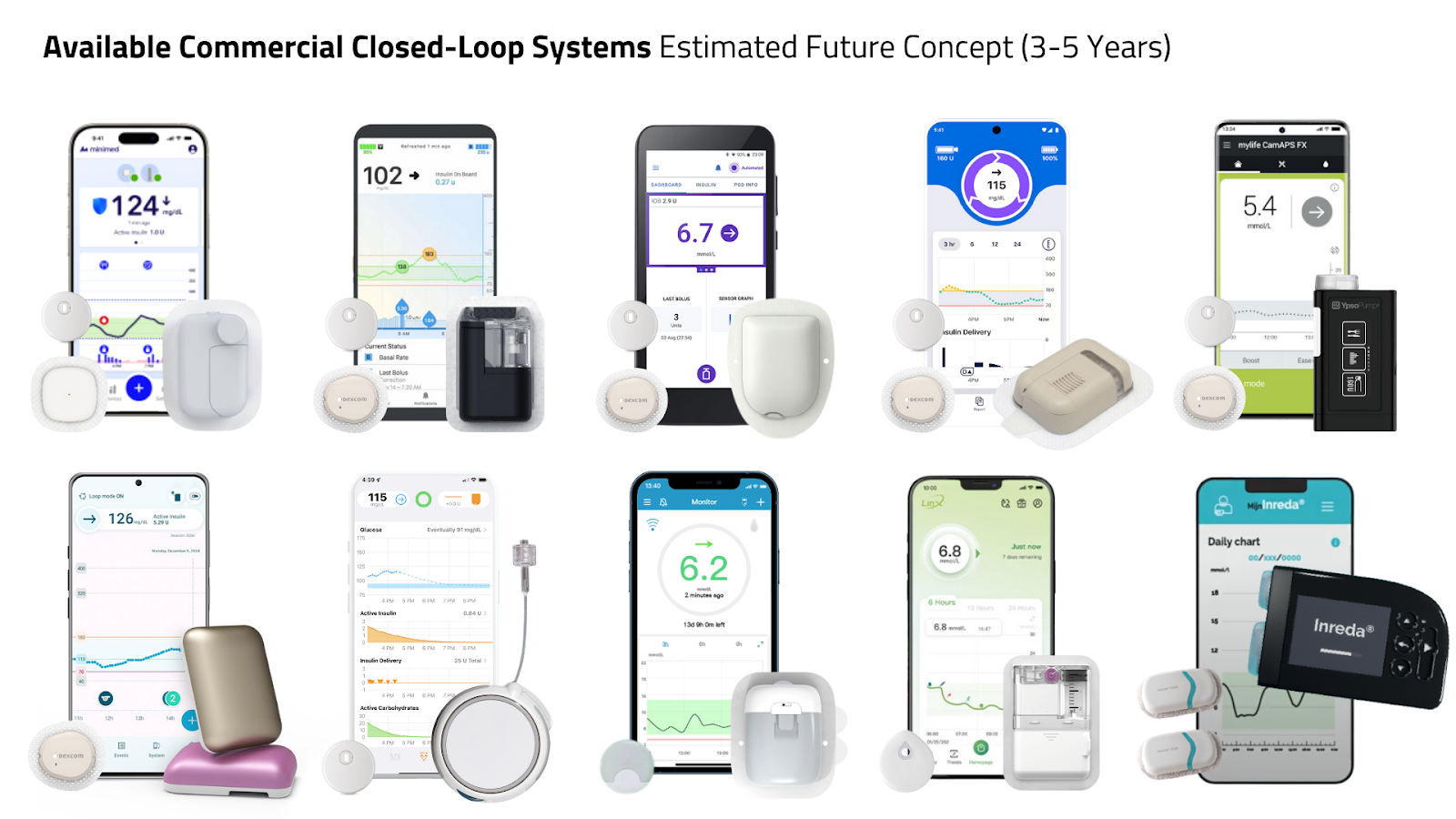
Automated Insulin Delivery options are shifting toward:
- patch pump options
- complete phone control
- and reduced need for meal boluses
AID is already standard of care in people with type 1 diabetes and increasingly preferred in people with type 2 diabetes on multiple daily injections, yet adoption still lags.
Systems are becoming simpler, more automated, and less dependent on user or provider input.
Competition is finally emerging, which should push usability forward and pricing downward.
This rapid evolution — combined with the increasing fragmentation of devices, algorithms, and indications — is exactly why we provide independent, accredited e-learning on every AID system.
Clinicians need a single, trusted place to stay up to date without losing time.
We offer:
- 9 free starter modules
- Full platform: €9.99/month
- Full platform: €95/year
>> Start now and stay ahead.
AID systems are here to stay — and your patients need them today.
Kind regards,